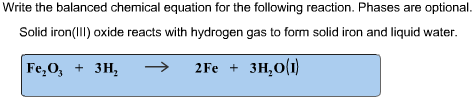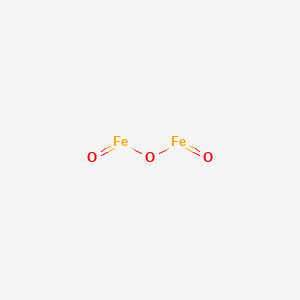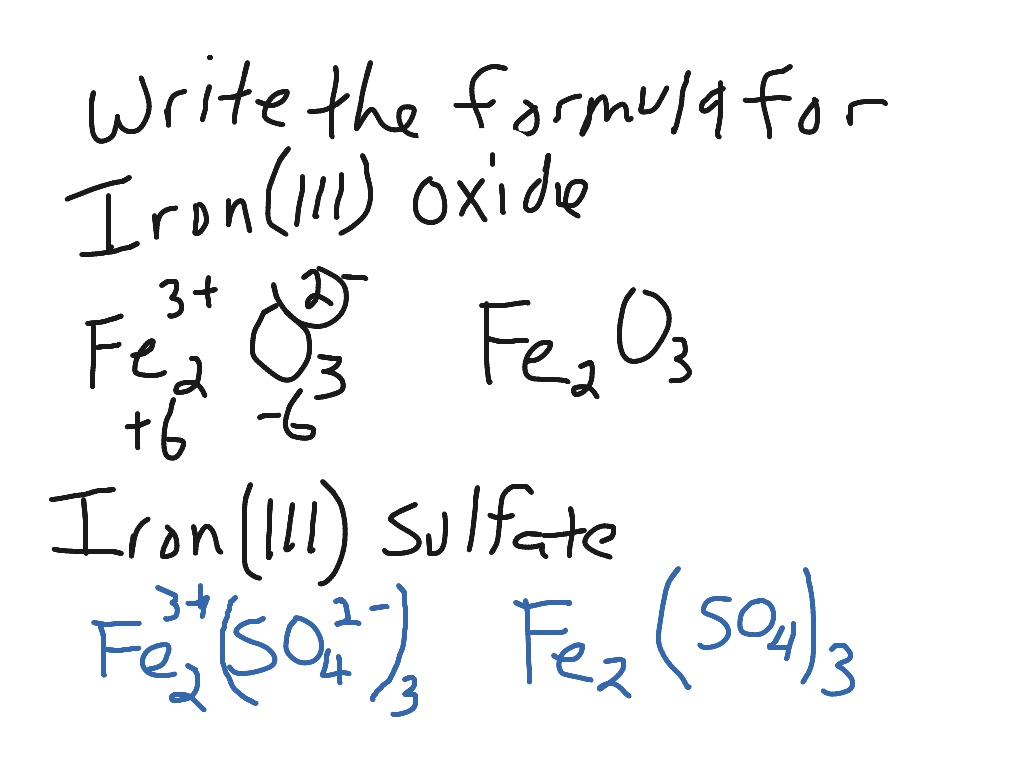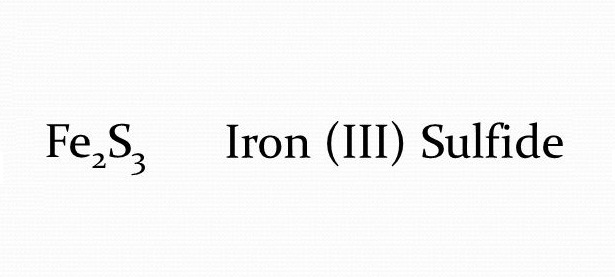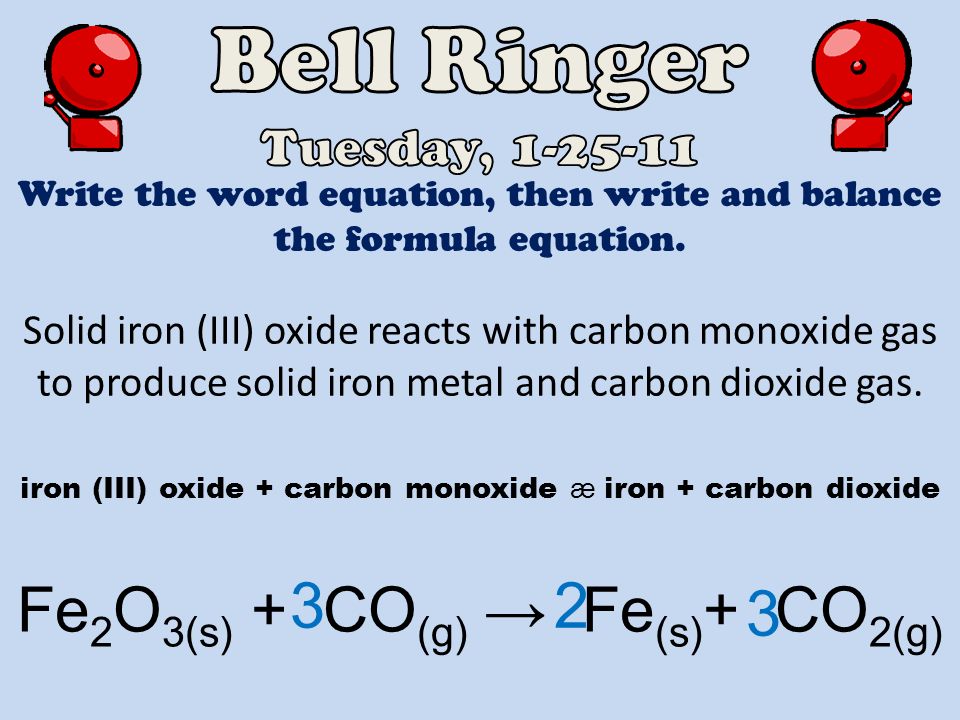
Tuesday January 25, 2011 (Types of Chemical Reactions; Predicting the Products of Chemical Reactions) - ppt download

Question Video: Writing the Equation for 𝐾_𝑐 in the Reduction of Iron(II) Ions by Silver Ions | Nagwa

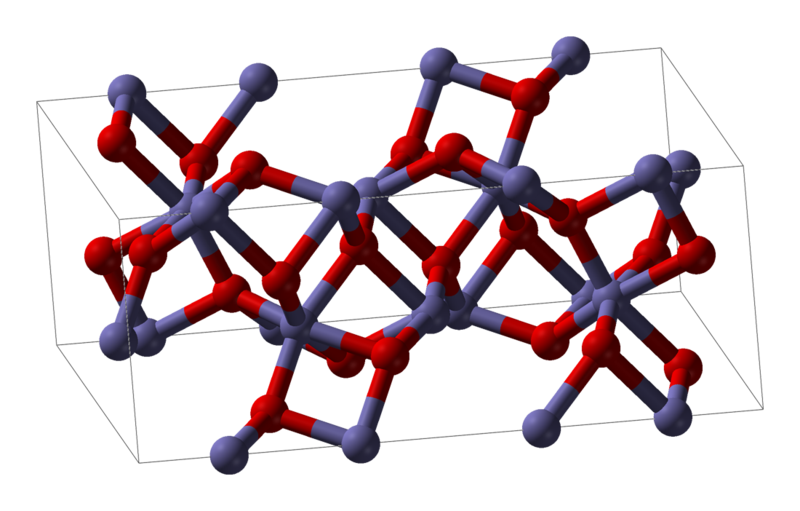
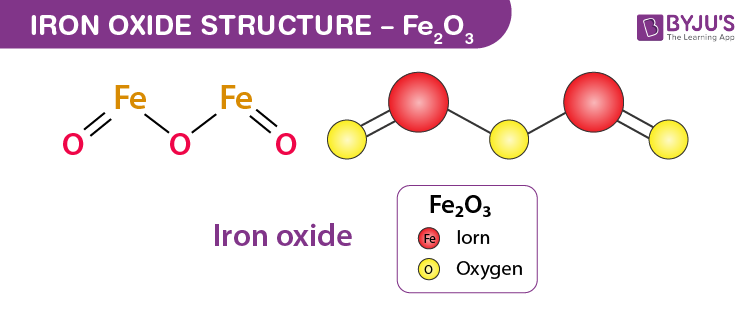
When Density Functional Approximations Meet Iron Oxides | Journal of Chemical Theory and Computation

A solid iron pole consists of a cylinder of height 220 cm and base diameter 24 cm, which is surmounted by another cylinder of height 60 cm and radius 8 cm. Find

SOLVED:Challenge Solid iron reacts with chlorine gas to form solid iron(lil chloride (FeCl,). Write the balanced equation and the equilibrium constant expression for the reaction.

SOLVED: Iron metal reacts with solid sulfur to produce solid iron(III) sulfide. Express your answer as a chemical equation. Identify all of the phases in your answer.