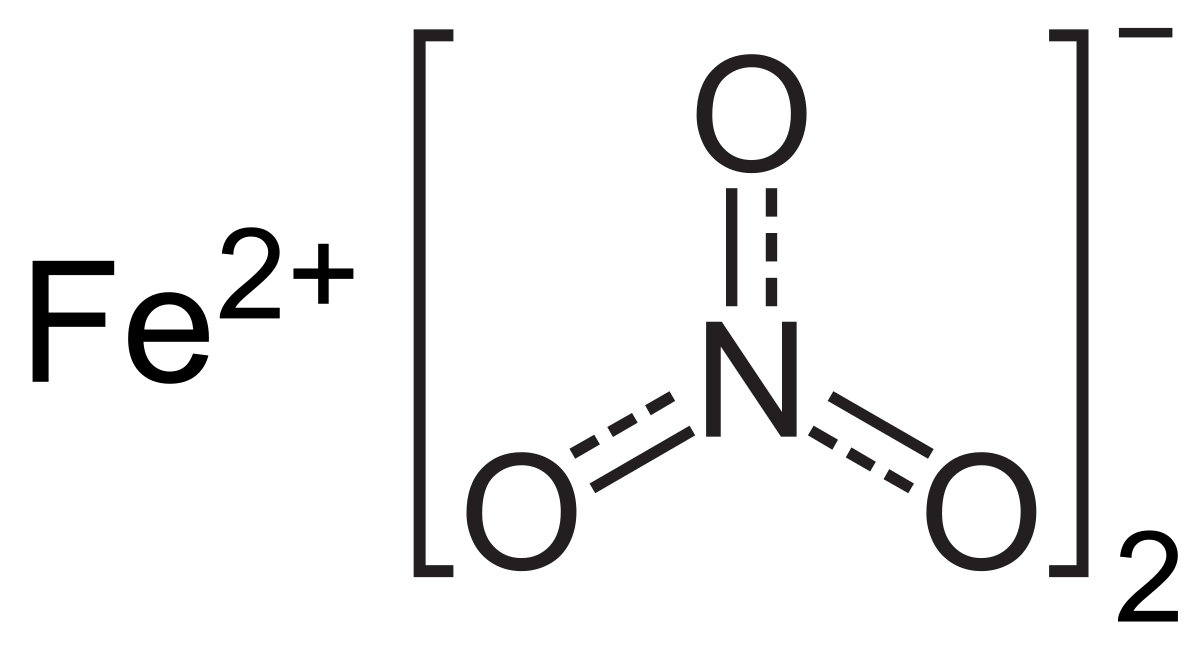
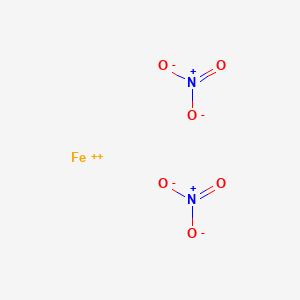
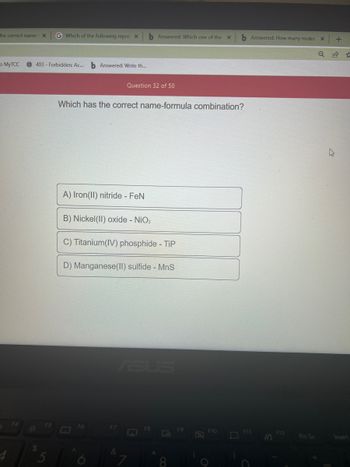
SOLVED: Which of the following correctly pairs the formula with the metal ion name? Select the correct answer below: Iron (II) nitride, FeN2 tin (IV) sulfide, SnS2 manganese (IV) nitride, MnN2 all
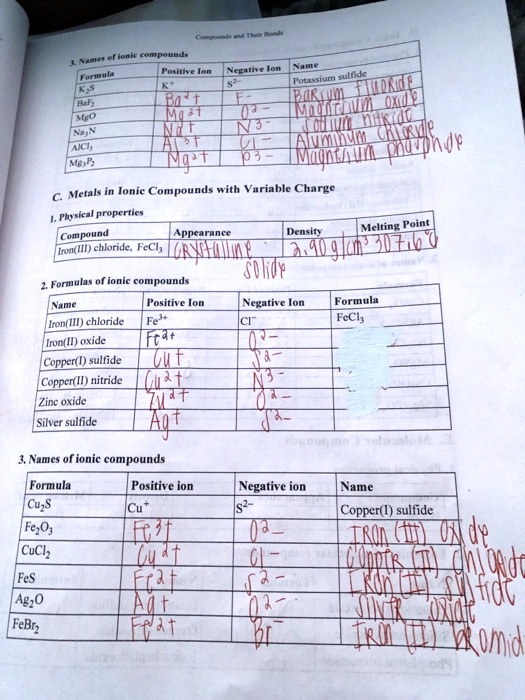
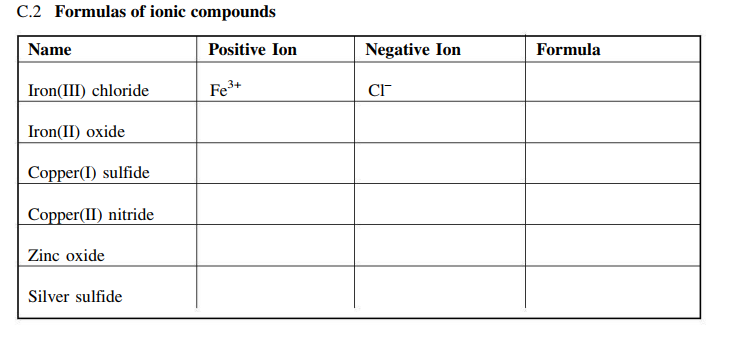
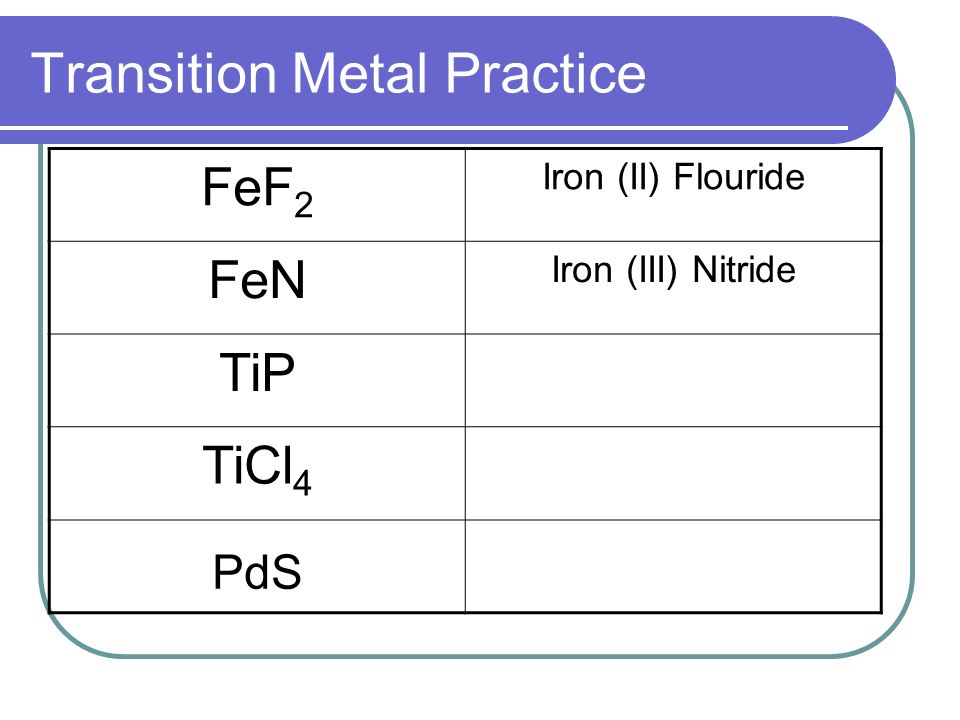
SOLVED: Text: Meet the Roman Empire's Final Fall Ncru(4lt Metals Ionic Compounds with Variable Charge Physical properties Melting Point Compound nnta Dcnellt Hh Iron(III) chloride; FcCl; SOWVp Formulas of ionic mnnnem Mume

_how-to-write-the-formula-for-iron-ii-nitride.webp)
_how-to-write-the-formula-for-iron-ii-nitride.jpg)


![Solved The compound [iron (II) nitride ] has the formula: | Chegg.com Solved The compound [iron (II) nitride ] has the formula: | Chegg.com](https://media.cheggcdn.com/study/840/8409396c-02f8-435b-8425-38f02201dfe0/image)