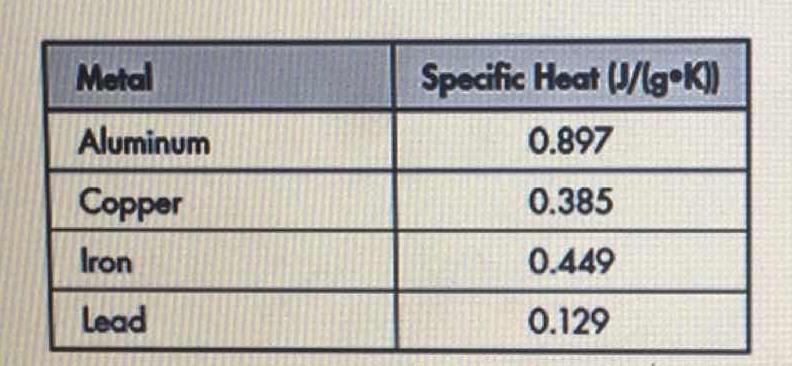
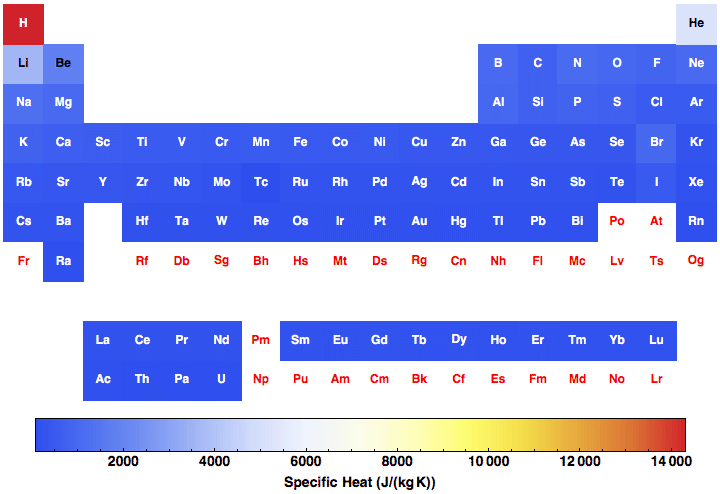
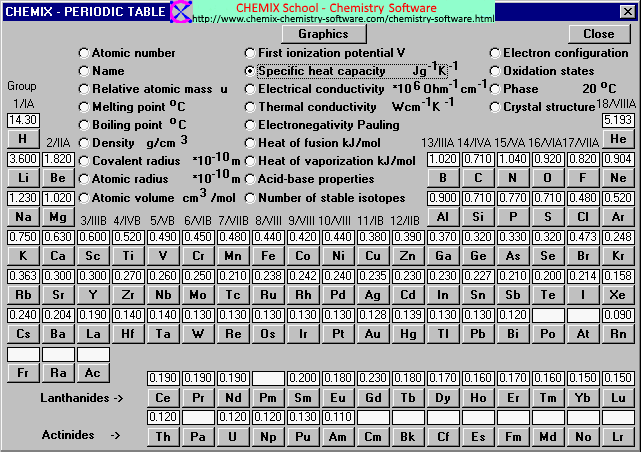
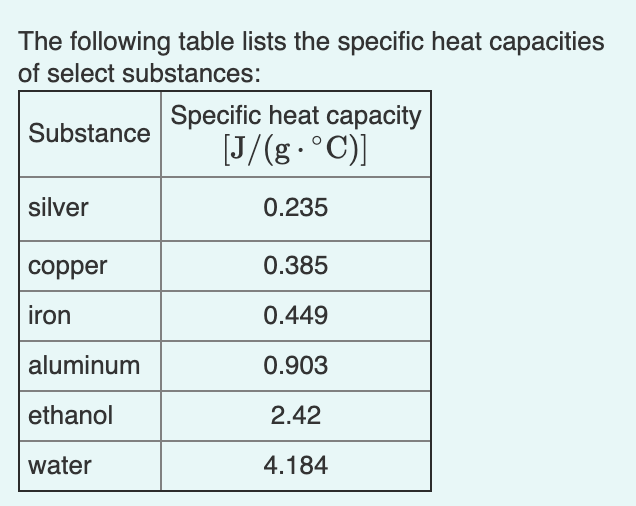
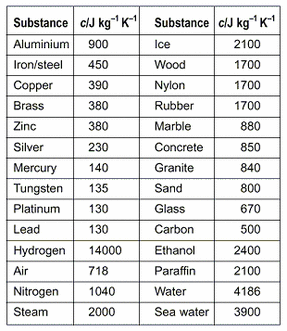
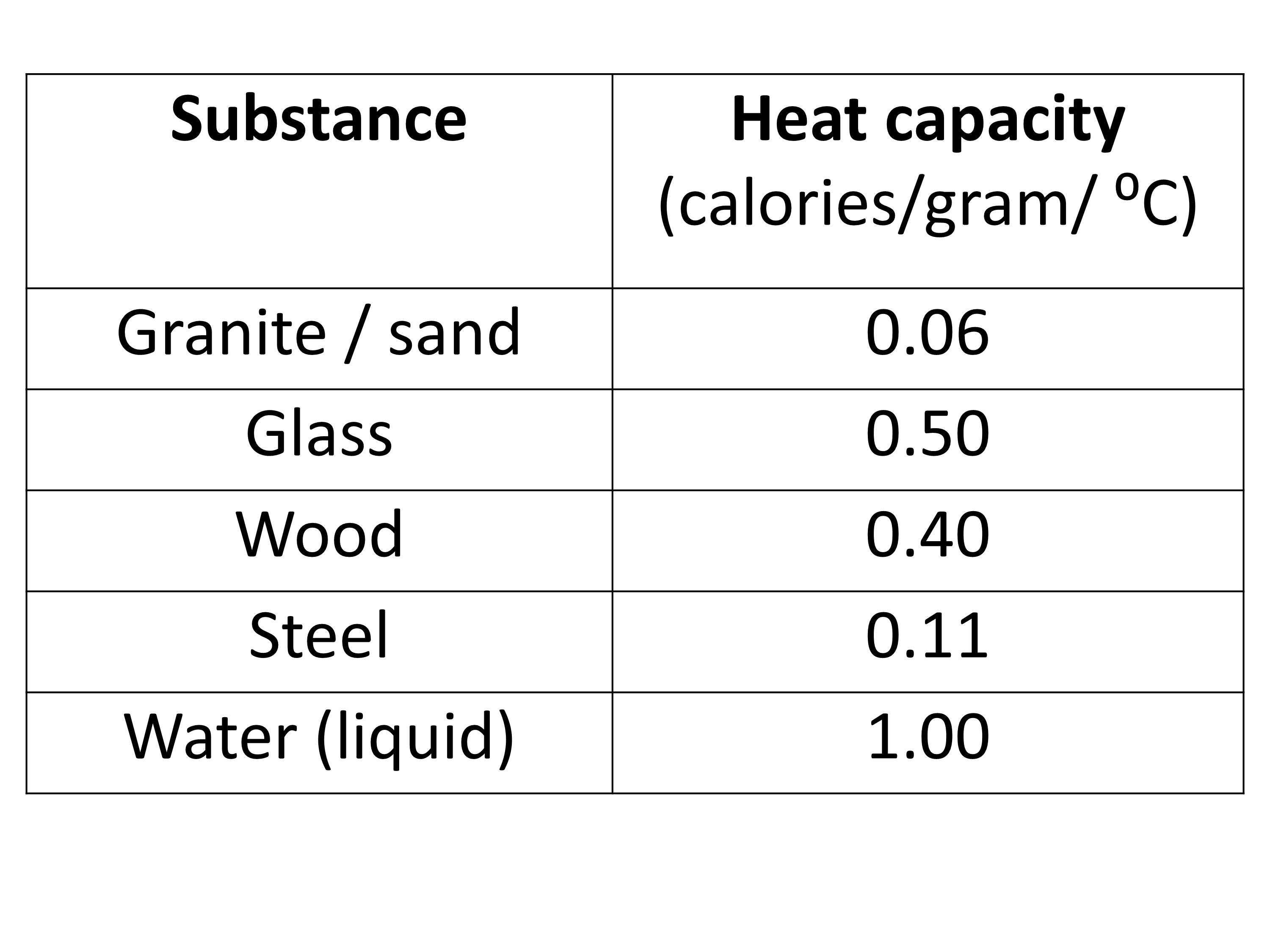
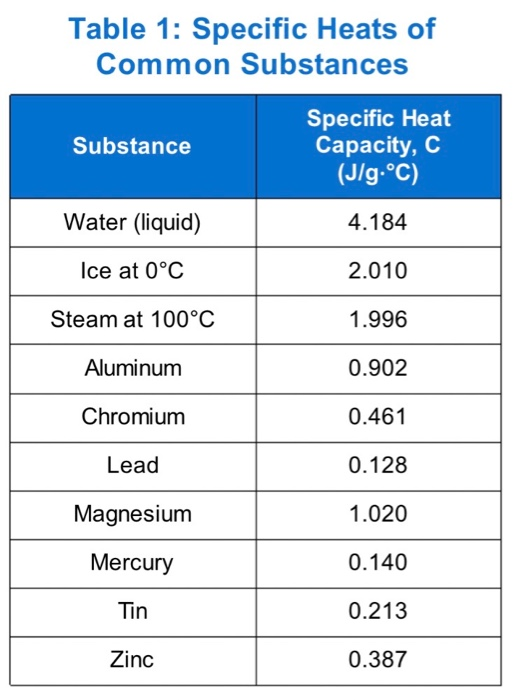
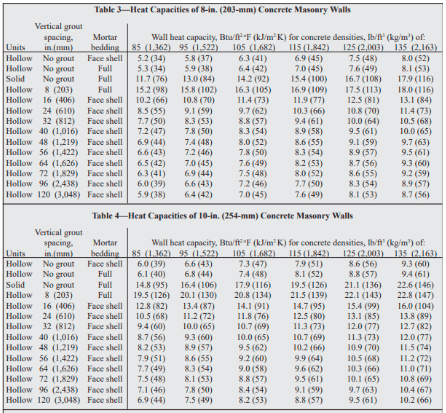
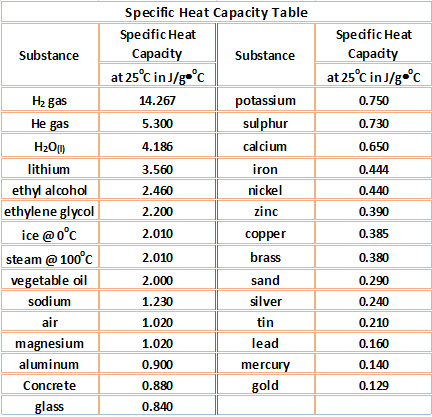
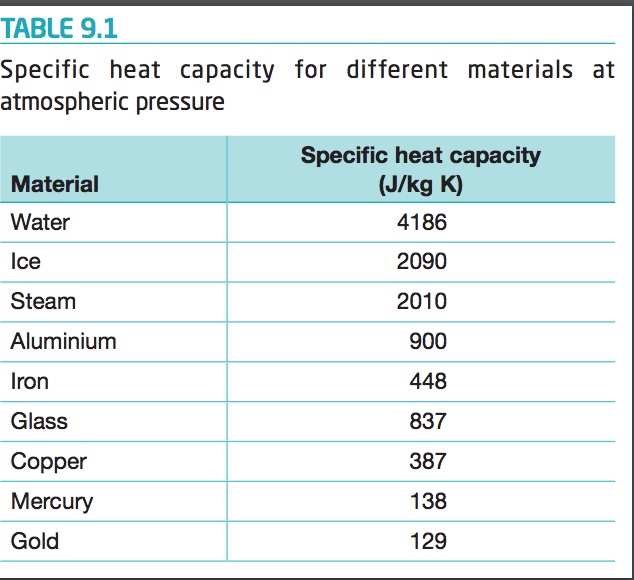
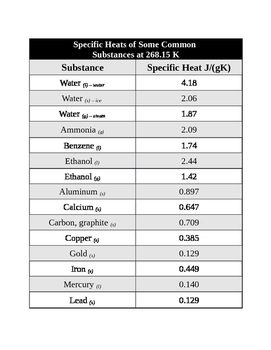
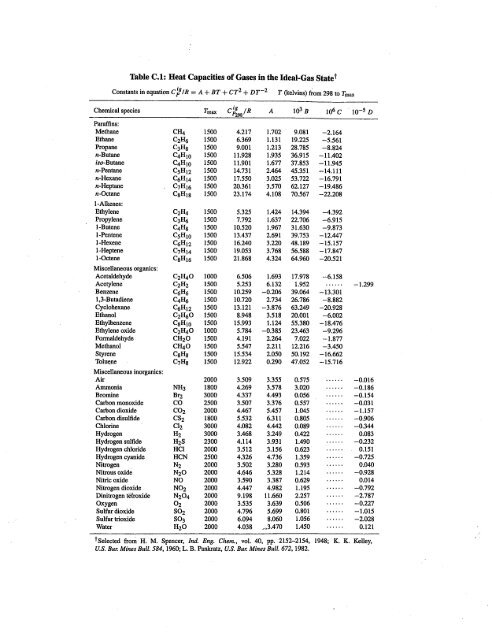
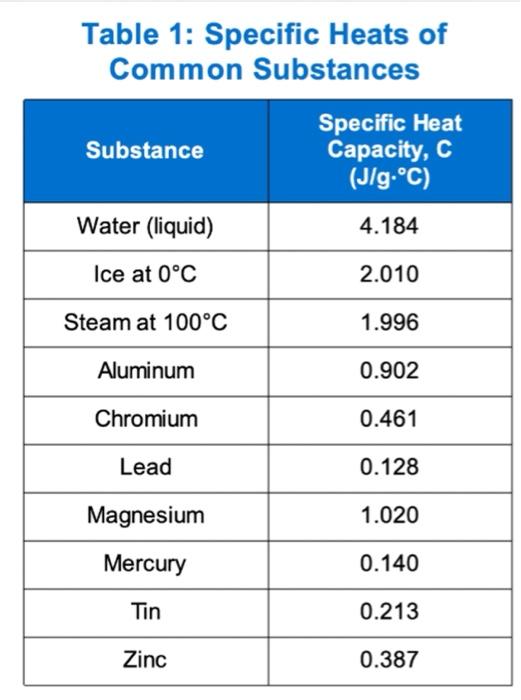
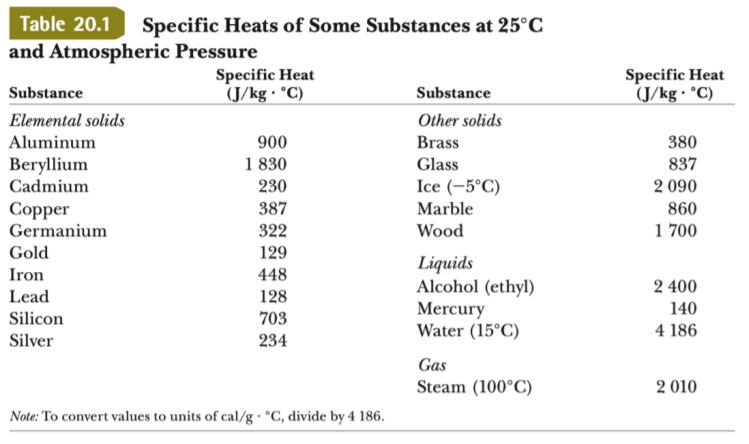
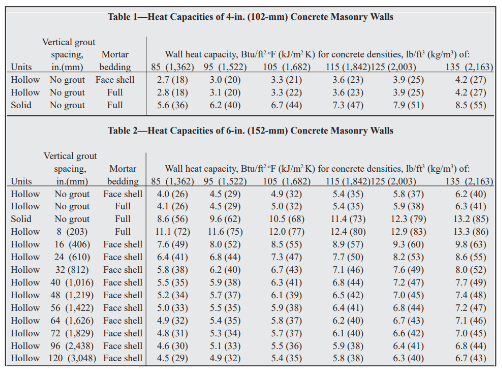
OneClass: Prepare a graph of specific heat capacities for metals versus their atomic weights. Combine...
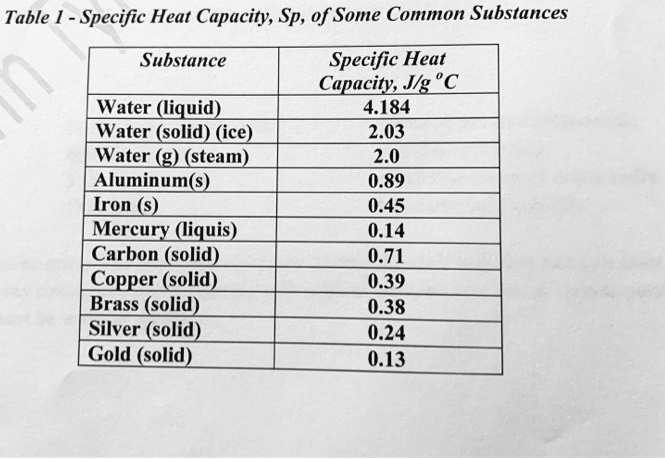
SOLVED: Table 1: Specific Heat Capacity, Sp, of Some Common Substances Substance Specific Heat Capacity, J/g 4.184 2.03 2.0 0.89 0.45 0.14 0.71 0.39 0.38 0.24 0.13 Water (liquid) Water (solid) (ice)

Table of Specific Heat Capacities: List of Thermal Conductivities | PDF | Molar Concentration | Temperature

Table 4 from Specific Heats Cv of Fluid Oxygen from the Triple Point to 300 K at Pressures to 350 Atmospheres. | Semantic Scholar
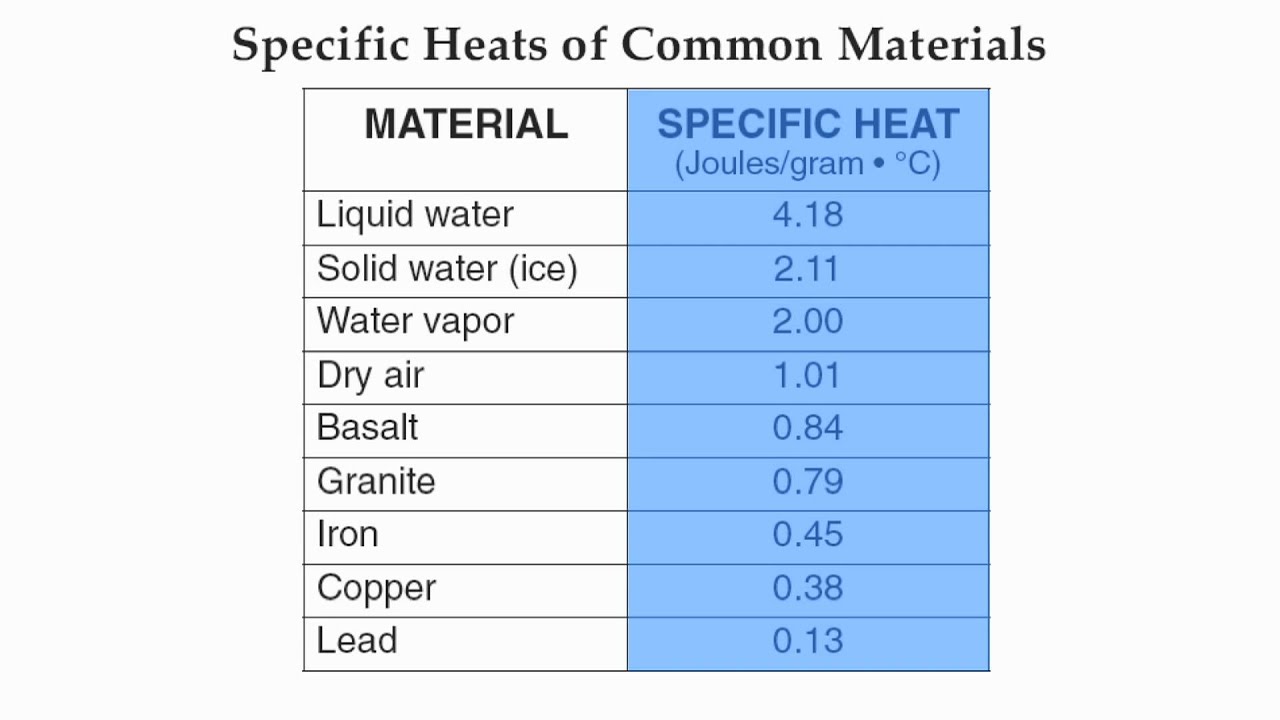
Reference Table Page 1-Specific Heat of Common Materials-Hommocks Earth Science Department - YouTube